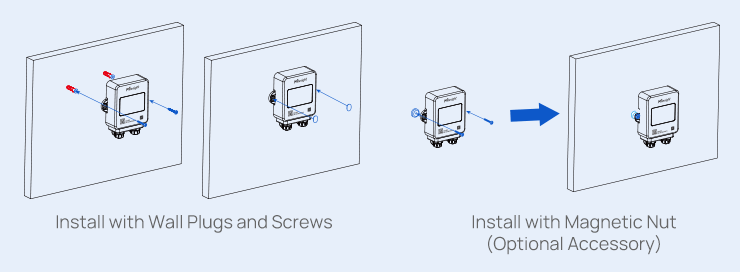
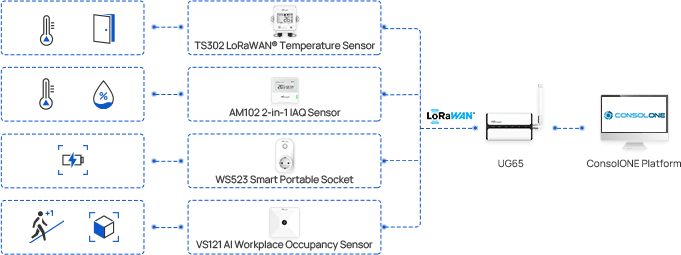
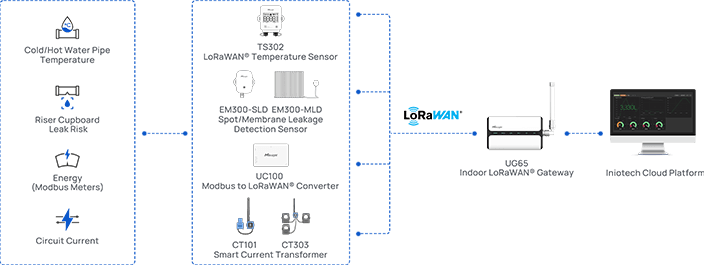
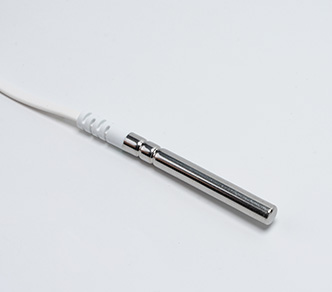
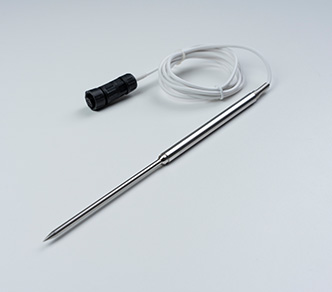
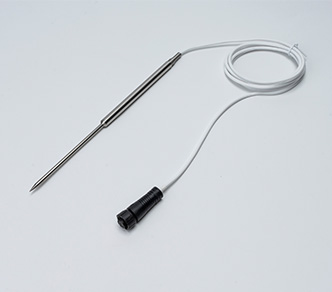
The TS30x supports regulatory compliance through a combination of accurate measurement, secure data handling, and automated recordkeeping.
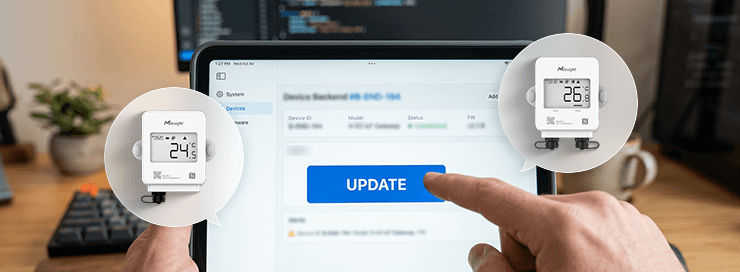
For 21 CFR Part 11 (ERES) compliance, the TS30x enables secure electronic records through encrypted data export in PDF format, ensuring that records are tamper-evident and audit-ready. Data is automatically logged without manual intervention, reducing the risk of gaps or human error that could compromise record integrity. When integrated with compatible platforms, the system also supports access controls and audit trail requirements critical to 21 CFR Part 11 adherence.
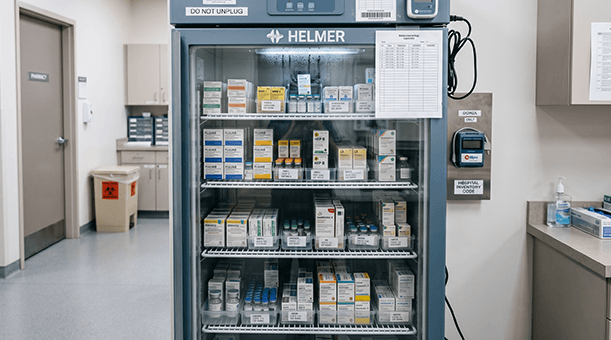
For HACCP compliance, the TS30x continuously monitors critical control points such as refrigerated storage, cold preparation areas, and food holding environments. Automated logging eliminates the reliance on manual temperature checks, while configurable threshold alerts ensure that any excursion is detected and can be acted on immediately. Historical records are stored and retrievable, providing the documentation needed to demonstrate that critical limits were consistently maintained.
Together, these capabilities help food and pharmaceutical operations reduce compliance risk, prepare for regulatory audits, and maintain the standards required to protect product quality and consumer safety.
 Discuss Your Needs
Discuss Your Needs